Introduction
The fundamental difference between single crystal, polycrystalline and amorphous solids is the length scale over which the atoms are related to one another by translational symmetry ('periodicity' or 'long-range order'). Single crystals have infinite periodicity, polycrystals have local periodicity, and amorphous solids (and liquids) have no long-range order.
- An ideal single crystal has an atomic structure that repeats periodically across its whole volume. Even at infinite length scales, each atom is related to every other equivalent atom in the structure by translational symmetry.
- A polycrystalline solid or polycrystal is comprised of many individual grains or crystallites. Each grain can be thought of as a single crystal, within which the atomic structure has long-range order. In an isotropic polycrystalline solid, there is no relationship between neighbouring grains. Therefore, on a large enough length scale, there is no periodicity across a polycrystalline sample.
- Amorphous materials, like window glass, have no long-range order at all, so they have no translational symmetry. The structure of an amorphous solid (and indeed a liquid) is not truly random - the distances between atoms in the structure are well defined and similar to those in the crystal. This is why liquids and crystals have similar densities - both have short-range order that fixes the distances between atoms, but only crystals have long-range order.
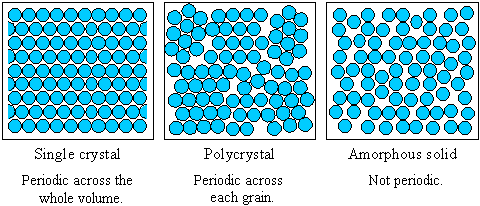
The range of crystalline order distinguishes single crystals, polycrystals and amorphous solids. The figure shows how the periodicity of the atomic structure of each type of material compares.
Many characteristic properties of materials, such as mechanical, optical, magnetic and electronic behaviour, can be attributed to the difference in structure between these three classes of solid.

