Barium titanate and phase changes
The temperature at which the spontaneous polarisation disappears is called the Curie temperature, TC.
Above 120°C, barium titanate has a cubic structure. It is therefore centro-symmetric and possesses no spontaneous dipole. With no spontaneous dipole the material behaves like a simple dielectric, such that its polarisation varies linearly with field. TC for barium titanate is 120°C.
Below 120°C, it changes to a tetragonal phase, with an accompanying movement of the atoms. The movement of Ti atoms inside the O6 octahedra may be considered to be significantly responsible for the dipole moment:
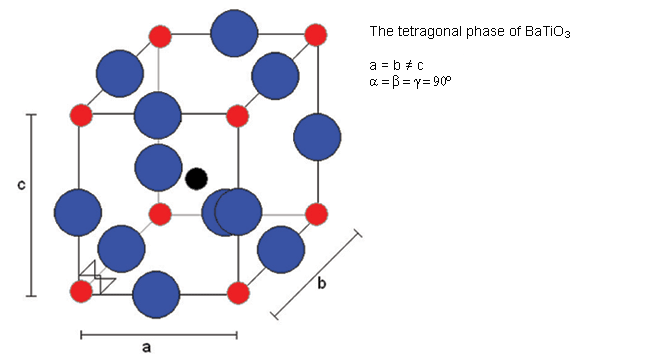
Cooling through 120°C causes the cubic phase of barium titanate to transform to a tetragonal phase with the lengthening of the c lattice parameter (and a corresponding reduction in a and b). The dipole moment may be considered to arise primarily due to the movement of Ti atoms with respect to the O atoms in the same plane, but the movement of the other O atoms (i.e. those O atoms above and below Ti atoms) and the Ba atoms is also relevant.
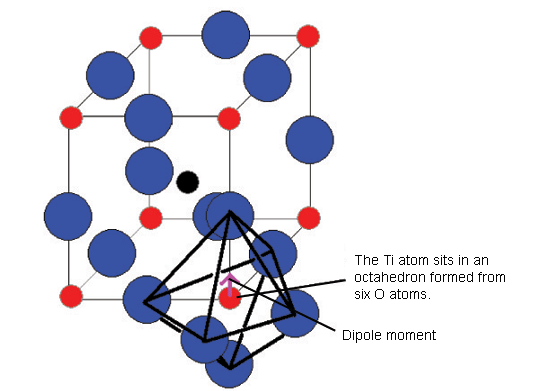
The diagram below shows the BaTiO3 structure with an O6 octahedron surrounding the Ti atom.
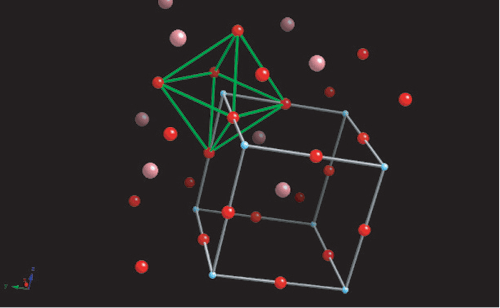
The switching to a cubic structure is the reason for the polarisation spontaneously disappearing above 120°C. Barium titanate has two other phase transitions on cooling further, each of which enhances the dipole moment:
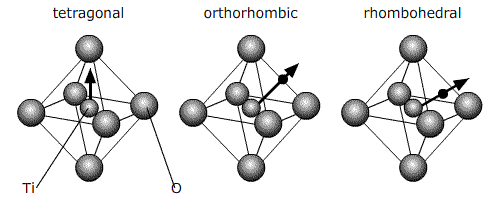
The phase which is reached after cooling to ~ 0°C from tetragonal is orthorhombic.
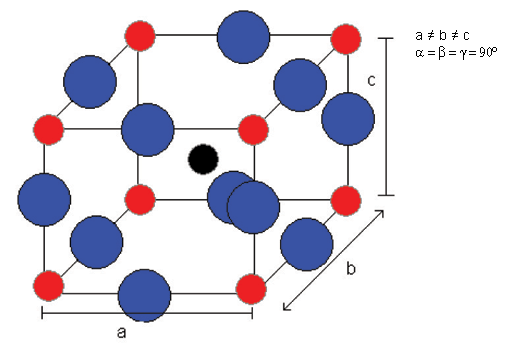
And then rhombohedral below -90°C:
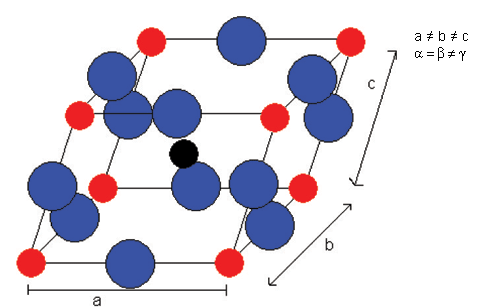
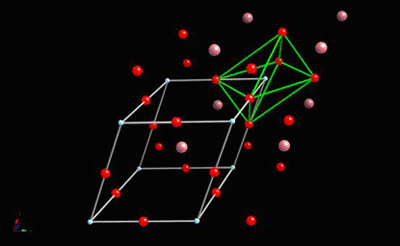
All of these ferroelectric phases have a spontaneous polarisation based to a significant extent on movement of the Ti atom in the O6 octahedra in the following way (using pseudo-cubic notation):