Theory 1
When we extend or compress a polymer elastically (i.e. there is no permanent deformation), we try to move the chain-ends apart or together. For a simple polymer chain to change its conformation, individual C-C bonds must twist from the trans to gauche position or vice versa - i.e. the torsion angles must change. This is a thermally activated process. At low temperatures, there is not enough thermal energy available to allow torsion angle changes, so the conformation becomes frozen in. The temperature above which the torsion angles can change is called the glass transition temperature. The changes in conformation also depend on time-scale, so the apparent value of Tg depends on the time-scale over which the behaviour is being monitored.
The strain in a polymer is accommodated by the change in shape of the individual molecules, but it should be noted that the response of the bulk polymer is influenced by the interactions between the molecules. This affects the ability of the bonds to rotate, and also the viscosity of the bulk polymer.
Therefore Tg depends on the polymer's architecture, and there are several factors influencing the transition:
Chain Length
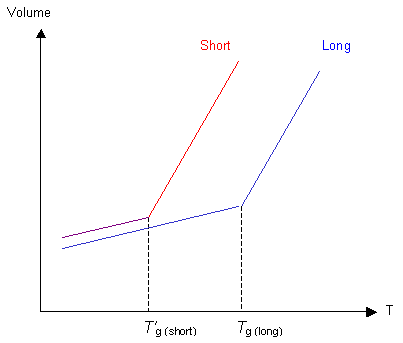
Each chain end has some free volume associated with it. A polymer with shorter chains will have more chain ends per unit volume, so there will be more free volume. Hence Tg' for shorter chains will be lower than Tg for long chains. Note that the shorter-chained polymer also has more free volume frozen in below Tg than the long-chained polymer.
Chain Flexibility
A polymer with a backbone that exhibits higher flexibility will have a lower Tg. This is because the activation energy for conformational changes is lower. Therefore, conformational changes can take place at lower temperatures.
Side Groups
Larger side groups can hinder bond rotation more than smaller ones, and therefore cause an increase in Tg. Polar groups such as Cl, CN or OH have the strongest effect.
Branching
Polymers with more branching have more chain ends, so have more free volume, which reduces Tg, but the branches also hinder rotation, like large side groups, which increases Tg. Which of these effects is greater depends on the polymer in question, but Tg may rise or fall.
Cross-linking
Cross-linking reduces chain mobility, so Tg will be increased. It also affects the macroscopic viscosity of the polymer, since if there are cross-links between the chains, then they are fixed relative to each other, so will not be able to slide past each other.
Plasticisers
Small molecules, typically esters, added to the polymer increase the chain mobility by spacing out the chains, and so reduce Tg.
Time Effects
The properties of an amorphous polymer above Tg can change with time
- At very short loading times the polymer can still be glassy because there is not time for the chains to move.
- At intermediate times the polymer may be rubbery - i.e. chains can uncoil and recoil between entanglements, which remain stable.
- At very long times, the chains can move past each other permanently, and so the polymer behaves as a viscous liquid.
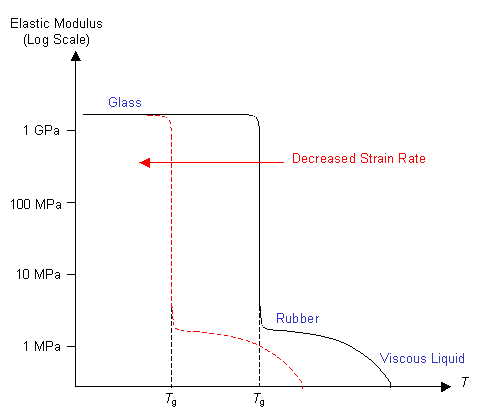
For a useful rubbery material it is necessary to suppress chain sliding. One way of doing this is to increase the amount of cross-linking in the polymer. In order for the chains to slide the cross-linking bonds must first be broken. So increasing the number of cross-links decreases the chains' ability to slide over each other. This extends the rubbery region at higher temperatures, so the graph looks like this:
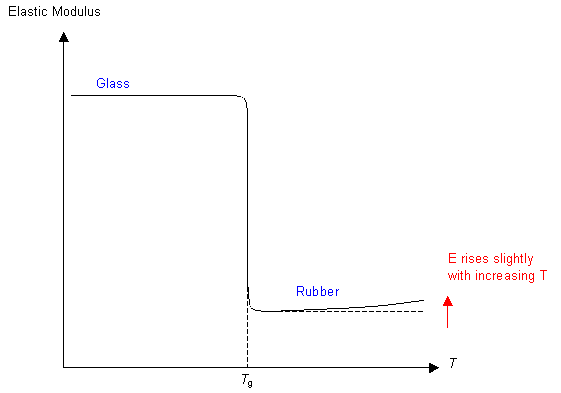
More about the slight rise in elastic modulus with increasing temperature in rubber is available in the Stiffness of Rubber TLP.

