Cross-Sections
To understand the rest of this TLP, it is vital to know about cross-sections.
What is a Cross-Section?
A cross-section quantifies the probability that a particle passing through a material will interact with the material. For example, a neutron absorption cross-section would quantify the probability that a neutron is absorbed as it travels through a material.
The following equation is a definition of the nuclear cross section σ
\[\sigma = \frac{C}{{N\delta .I}}\]
For neutrons passing through a plate of thickness δx (m), C is the number of events occurring per unit area (m−2), N is the number of nuclei per unit volume, or nuclear number density (m−3), and I is the number of neutrons passing through a unit area (m−2). As the behaviour depends on neutron energy, the cross-section must be specified for neutrons of a given energy (i.e. monoenergetic).
The Nδx term is often grouped together, since when multiplied by σ it is equal to C / I, a dimensionless quantity that is the probability of a neutron interacting, i.e. the ratio of the number of events occurring per unit area to the number of neutrons travelling through that same area.
Types of Cross-Section
Several different cross-sections will be mentioned in this TLP. Standard notation is used below, where (a,b) means an atomic interaction in which a is absorbed and b is emitted.
Elastic scattering (n,n): the cross-section of a neutron undergoing elastic scattering by a nucleus The total kinetic energy of the neutron and the nucleus is the conserved. Any energy that the neutron loses is due to the nucleus recoiling after the neutron is scattered.
Inelastic scattering (n,n'): a neutron is briefly absorbed by a nucleus, leaving it in an excited state. The nucleus can later return to its ground state, losing its excess energy as a gamma ray.
Radiative capture (n,γ): a neutron is absorbed by a nucleus, which gives out a gamma ray as a result.
Fission (n,f): neutron causes a nucleus to split into fragments and more neutrons.
Alpha decay (n,α): neutron causes a nucleus to lose two protons and two neutrons in the form of a helium nucleus. This interaction is important when considering the transmutation of elements, and how radioactivity is induced in a material.
Virtually any possible interaction has its own specific cross section; the ones above are just some of the most common. Other important interactions include (n,p) and (n,2n).
Cross-Section and Neutron Energy
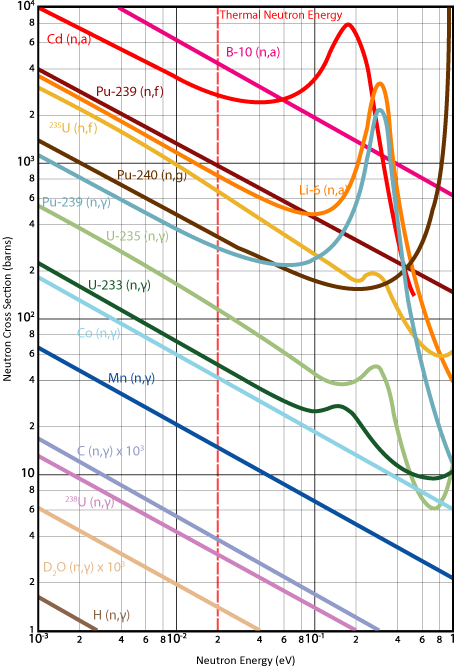
Graph showing neutron cross-section against neutron energy. [Adapted from graph by Napy1Kenobi CC[BY][SA], source data unknown]
As the log-log graph above shows, cross-sections vary with neutron energy. Since most neutrons are in the thermal range (about 0.025 eV, or about 4 × 10−21 J), cross-sections are often quoted for this neutron energy.
Even though cross-sections do vary with energy, nuclides still have characteristically "high" or "low" cross sections. For example, as the graph shows, 235U (n,γ) has a higher cross section than 233U (n,γ) over almost all energy ranges.
The peaks in the graph are due to resonance effects. The reasons for these are beyond the scope of this TLP.
The Macroscopic Cross-Section
So far we have examined the microscopic cross-section. When talking about actual materials, the macroscopic cross-section is more commonly used.
Each element present in a material has its own macroscopic cross-section (m−1) defined by the following equation, where N is the nuclear number density as used earlier (m−3).
\[\Sigma_{i} = N_{i}\sigma_{i}\]
And for the material as a whole, its macroscopic cross-section is therefore:
\[\Sigma = N_{1}\sigma_{1} + N_{2}\sigma_{2} + \cdot \cdot \cdot + N_{i}\sigma_{i} + \cdot \cdot \cdot\]
The macroscopic cross-section is the probability that a neutron will undergo a reaction per unit path length travelled in the material.
The probability that a neutron travels a distance x without interacting therefore is:
$$\exp(-\Sigma x)$$
And the neutron mean free path, i.e. the average distance a neutron travels before interacting, can be found by integrating over this quantity as follows:
$$\lambda = \int_0^\infty x {\rm{P}}(x){\rm{d}}x = \int_0^\infty x \Sigma \exp ( - \Sigma x){\rm{d}}x = {1 \over \Sigma }$$
Interactive Graph of Macroscopic Cross Section
Try out the graph below to see what effect mass, density and microscopic cross-section have on the macroscopic cross-section. The nuclear number density is calculated by simply working out the number of nuclei present in the material given the molar mass and its density. This method makes the approximation that all the mass is present as nuclei, which is true to a reasonable degree of accuracy (electrons also have mass, but are only about 1/2000 the mass of a single nucleon and so do not contribute significantly).
The graph is editable: double-click on a cell to edit the numbers given. The arrows along the x-axis show the mean free path of the neutron through the material.

