Corrosion Control
There are several ways kinetics can be employed to reduce or prevent corrosion.
Barriers and coatings
A barrier can be employed to prevent the electrolyte coming into contact with the metal.
Tin is the usual barrier, as it does not react in most aqueous solutions. It is used in food cans and has the useful property that if the barrier fails, then the steel in the can corrodes. If zinc were used instead it would begin to act as a sacrificial anode should the barrier fail. This would protect the steel but the distinct disadvantage is that hydrogen is evolved at the steel cathode and if it were to build up inside a closed container an explosive situation could arise.
Other inert metals may be used as barriers, as can polymers, ceramics and paint.
Anodic protection
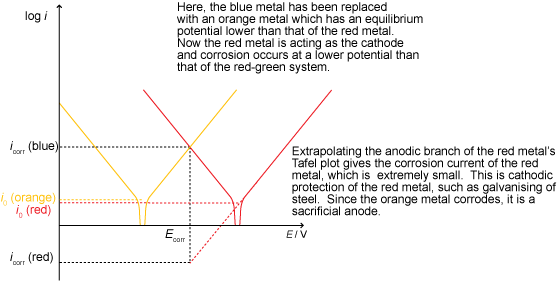
Anodic protection involves raising the potential of the metal in order to develop a passivating layer (such that protection is due to inhibited kinetics).
Sodium carbonate, a base, acts to remove the acidity in the solution and drives the reaction towards the right of the Pourbaix diagram. Above a certain pH, metals tend to form a passive layer as a passive species is stable under these conditions.
Potassium chromate works by providing a source of chromate ions that penetrate the surface of the metal, forming a stable, passive chromium oxide layer on the surface of the anode and thus prevents corrosion by forming a passivation layer in a similar way to that seen in stainless steels.
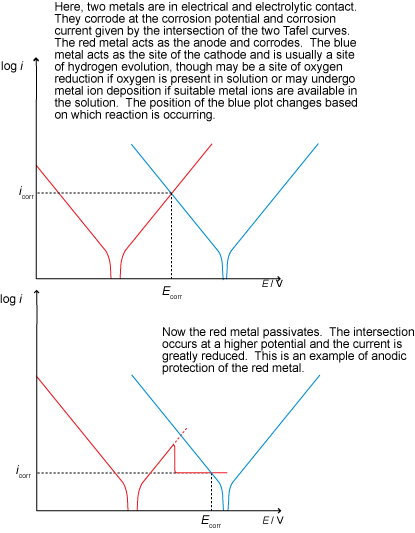
Cathodic protection
Cathodic protection involves lowering the potential of the metal in order to make it more thermodynamically stable. This may be done using an impressed current (a supply of electrons from an external source) or with a sacrificial anode, as shown below: