Single crystals: Shape and anisotropy
A single crystal often has distinctive plane faces and some symmetry. The actual shape of the crystal will be determined by the availability of crystallising material, and by interference with other crystals, but the angles between the faces will be characteristic of the material and will define an ideal shape. Single crystals showing these characteristic shapes can be grown from salt solutions such as alum and copper sulphate.
Gemstones are often single crystals. They tend to be cut artificially to obtain aesthetically pleasing refractive and reflective properties. This generally requires cutting along crystallographic planes. This is known as cleaving the crystal. A familiar example is diamond, from which decorative stones can be cleaved in different ways to produce a wide range of effects.
To see a variety of symmetrical naturally formed minerals, visit the mineral galleries website.
Consider the following three-dimensional shapes:
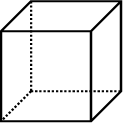 |
Cube: 6 identical squares |
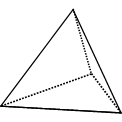 |
Tetrahedron: 4 identical equilateral triangles |
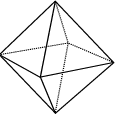 |
Octahedron: 8 identical equilateral triangles |
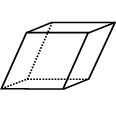 |
Rhombohedron: 6 identical parallelograms with sides of equal length |
You can make your own cube, octahedron and tetrahedron by printing the following pages and following the instructions on them.
These three shapes are the most important in materials science, and you should be very familiar with them!
The symmetry exhibited by real single crystals is determined by the crystal structure of the material. Many have shapes composed of less regular polyhedra, such as prisms and pyramids.
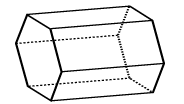 |
Hexagonal prism: 2 hexagons and 6 rectangles |
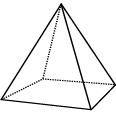 |
Square-based pyramid: 4 triangles and a square |
Not all single crystal specimens exhibit distinctive polyhedral shapes. Metals, for example, often have crystals of no particular shape at all.
 |
These quartz specimens show a range of shapes typically exhibited by crystals. (Click on an image to see a larger version.)
Most single crystals show anisotropy in certain properties, such as optical and mechanical properties. An amorphous substance, such as window glass, tends to be isotropic. This difference may make it possible to distinguish between a glass and a crystal. The characteristic shape of some single crystals is a clue that the properties of the material might be directionally dependent. The properties of polycrystalline samples can be completely isotropic or strongly anisotropic depending on the nature of the material and the way in which it was formed.



