Re-use of this resource is governed by a Creative Commons
Attribution-
NonCommercial-ShareAlike 4.0 International
https://creativecommons.org/licenses/by-nc-sa/4.0/
NonCommercial-ShareAlike 4.0 International
https://creativecommons.org/licenses/by-nc-sa/4.0/
Close packed structures

To build a close packed structure we first consider the
closest packed structures in two dimensions
closest packed structures in two dimensions
Either upon sites equivalent to this point
There are two distinct ways we can place a second layer
upon this layer
upon this layer
Or upon sites equivalent to this point
We will now refer to the bottom (red) layer as A
and the
second (orange) layer as B
second (orange) layer as B
For the third layer, again there are two distinct ways
that it
can be placed. However, this time where we place it will
determine which sort of structure we get.
can be placed. However, this time where we place it will
determine which sort of structure we get.
Third layer can be placed on sites equivalent to this
point X
Or upon sites equivalent to this point Y
Or upon sites equivalent to this point Y
Notice that the X sites
are positioned above atoms from layer A
while the Y sites are positioned above gaps in the structure.
while the Y sites are positioned above gaps in the structure.
This structure has the form ...ABA... That is, every
A layer is followed by a B layer and every B layer
is followed by an A layer
The unit cell has a hexagonal lattice type so this close
packing type is called hexagonal close packing or hcp
This structure is usually represented by the following
unit cell.The unit cell has a hexagonal lattice type so this close packing
type is called hexagonal close packing or hcp
If the atoms of the third layer are, instead, placed over
the Y sites then the atoms will not coincide with any other layer.
The third
layer is then labelled C.
layer is then labelled C.
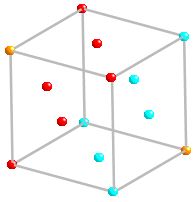
The form of this close packed structure is ...ABC...
It is a cubic close packed or ccp structure. The unit
call used is a face centered
cubic (fcc) unit cell.
cubic (fcc) unit cell.
For an explanation as to why the fcc unit cell is used
press Play
Play
To continue press Next
Play
To continue press Next
The form of this close-packed structure is ...ABC....
It is a cubic-close-packed or ccp structure. The unit
cell used is a face centred
cubic (fcc) unit cell.
cubic (fcc) unit cell.
These spheres are arranged in ABC layers
Here the close packed layers are coloured and labelled
as before
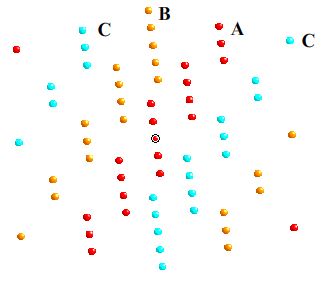
Here the close packed layers are coloured and labelled
as before
Notice that we can apply a cubic unit cell to the structure
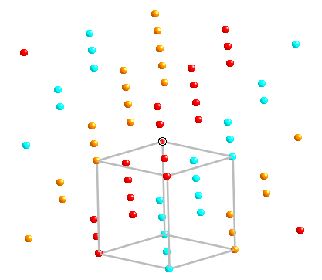
Notice that we can apply a cubic unit cell to the structure
The unit cell is face centred cubic