Mechanisms of Radiation Damage 1
Most of the radiation damage in a reactor is from the neutron flux being produced in the core. Other forms of radiation, such as gamma radiation, are very weakly interacting and don’t produce much effect. The principles in this section can in theory apply to any material, but the key materials are steels (e.g. a cold-worked 316 stainless steel).
Transmutation – (n, α) – Production of Helium
As seen in the previous section, there are several ways in which neutrons can interact with nuclei, including absorption of the neutron by the nucleus, making the nucleus unstable so that it decays, releasing an alpha particle in the process. Alpha particles consist of two protons and two neutrons, i.e. a 4He nucleus. Since they are 2+ positively charged, they are very highly ionizing, and will they quickly pick up electrons from the surrounding lattice and become elemental helium.
In stainless steels, the (n, α) interaction does not occur often with iron itself, but is mostly as a result of the nickel content of the alloy, as the graph of its cross section below shows.
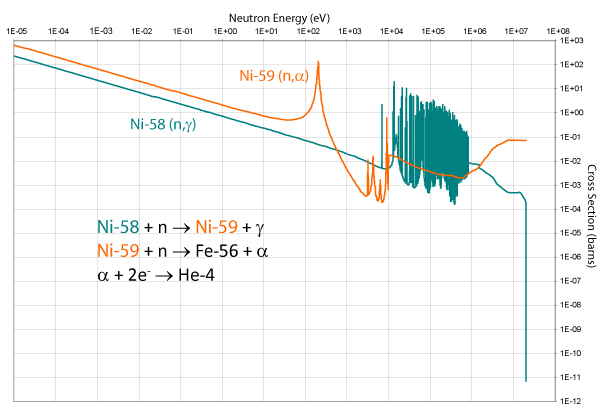
The presence of helium in the metal causes embrittlement and can act as a nucleation point for voids, which can lead to swelling.
Additionally, the neutron flux can induce further radiation. This occurs when a neutron transmutes an element into a radioactive one. This is undesirable, because it creates more low-level radioactive waste to contain when the reactor is eventually decommissioned.
Frenkel Defects
There are many proposed mechanisms of radiation damage, but on a fundamental level a single neutron scattering event can be considered.
If a neutron of sufficient energy scatters off a nucleus, the nucleus itself is displaced. The atom associated with the nucleus finds itself embedded into the structure elsewhere in a high-energy, interstitial site. It is termed a self-interstitial as the matrix and interstitial atoms are in principle the same. The site the atom previously occupied is now empty: it is a vacancy. In this way, self interstitial-vacancy pairs are formed, and these are called Frenkel defects.
Threshold Energy
At lower energies, the neutron collision causes the nucleus to vibrate, but the nucleus is not displaced. The excess energy is dissipated through the lattice as heat. The threshold energy to form a Frenkel defect depends on the nuclei present and the structure of the material (e.g. the phase of iron). It is typically in the range 10–50 eV (2–8 × 10−18 J). Note that when the neutron scatters off a nucleus, not all of its energy is transferred. This means that the minimum kinetic energy of the neutron is be larger than this threshold value, typically by a factor of 2–3.
This threshold energy is commonly given the symbol Ed. It is the energy required to overcome the potential barrier to move from one lattice site to another, and is approximately twice Es, the energy of sublimation, since twice as many bonds are broken to move an atom within a lattice as removing it from its surface, plus a contribution of 4–5 Ec, where Ecis the energy loss by electron stopping (required to allow the lattice to relax after the atom has been displaced).
Displacement Spikes
Neutron scattering events are not isolated. On average, each displaced atom might then go on to displace further atoms, and likewise the neutron that caused the first displacement might go on to displace further atoms. This means that there is a local cascade of displacements, known as a displacement spike, within which there is a large amount of disorder in the structure. This is illustrated with a simulation, below:
The Kinchin and Pease Model
A neutron scattering from an atom imparts an energy Ep to it. This primary knock-on atom (PKA) with energy Ep then displaces other atoms, ultimately giving a displacement cascade if Ep is high enough. The number of atoms displaced by the PKA is difficult to calculate, but a simple model (attributed to Kinchin and Pease) can capture much of the basic physics. The assumptions are:
- the cascade is a sequence of two-body elastic hard-sphere collisions;
- a minimum energy transfer Ed is required for displacement;
- the maximum neutron energy available for transfer is the cut-off energy Ec, set by loss to the electrons (electron stopping);
- the atoms are randomly distributed, so that channelling and other effects of crystal structure are ignored.
A full derivation can be found in Fundamentals of Radiation Science by Gary Was. The average number of atoms displaced by a PKA of energy Ep is:
0 for Ep< Ed
1 forEd < Ep< 2Ed
Ep/2Ed for 2Ed < Ep<Ec
Ec/2Ed for Ep ≥Ec

