Micrograph Library
Browse the libraryAdvanced searchSystemsCompositionsTechniquesKeywordsPhase diagramsHelpPreferencesAbout the micrograph libraryTerms of useContribute micrographs!FeedbackLinksCredits Print this page
Full Record for Micrograph 657

[122 KB]
View micrograph
.. in new window
View micrograph and record
.. in new window
You can also view and download the micrographs on Flickr
- Micrograph no
- 657
- Brief description
- Al-4 wt% Cu, age hardened alloy
- Keywords
- aged
 , alloy
, alloy  , aluminium
, aluminium  , copper, hardening
, copper, hardening  , heat treatment
, heat treatment  , metal, quenching
, metal, quenching  , vacancy
, vacancy - Categories
- Metal or alloy
- System
- Al-Cu
- Composition
- Al 96, Cu 4 (wt%)
- Standard codes
- Reaction
- Processing
- Age hardening, solution heat treatment
- Applications
- Aerospace applications, including struts and panels, making use of the high specific strength and stiffness.
- Sample preparation
- Etched in sodium hydroxide solution
- Technique
- Reflected light microscopy
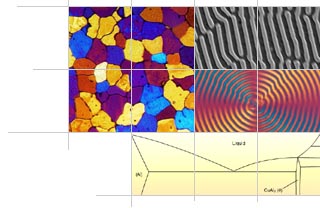
- Length bar
- 20 μm
- Further information
- This alloy was solution treated. Hardening is achieved by the controlled rejection of copper from a supersaturated solid solution. There is some additional hardening from precipitates such as MgSi2. From the phase diagram for the pure aluminium-copper binary system, it can be seen that the solubility of copper in -aluminium increases with increasing temperature up to the eutectic temperature of about 550°C. The equilibrium microstructure below the eutectic temperature is a two-phase mixture of -aluminium and the Al2Cu intermetallic phase (also known as q phase). The initial solution heat treatment aims to obtain the maximum possible concentration of copper in solution. Rapid quenching from the solution temperature prevents the kinetically slow precipitation of , forming a highly supersaturated solid solution of copper.
Rapid quenching also preserves the large number density of vacancies in the aluminium lattice from the high solution temperature. This increases copper diffusion rates at low temperature and accelerates ageing. Care must be taken with commercial alloys where the additional alloying elements reduce the eutectic temperature. This reduces the maximum solution heat treatment temperature since heating above the eutectic temperature causes the growth of a brittle intergranular eutectic.
Ageing may occur at room temperature (Natural Ageing) or above (Artificial Ageing), and takes place via a sequence of precipitation reactions. At low temperatures, the sequence may not be completed even after very long times, whereas at high temperatures, where diffusion is rapid, the early stages may not be observed.
The microstructure in this sample shows q phase at high magnification. The crystallographic relationship between the precipitates and the aluminium lattice is apparent from the alignment of the precipitates. - Contributor
- Dr J Marrow
- Organisation
- Department of Materials Science, University of Manchester and UMIST
- Date
- 07/11/02
- Licence for re-use
 Attribution-NonCommercial-ShareAlike 4.0 International
Attribution-NonCommercial-ShareAlike 4.0 International

