Batteries
AimsBefore you startIntroductionBasic principlesThermodynamics and kineticsPrimary batteriesZinc/carbon batteriesAlkaline/manganese oxide batteriesZinc/silver oxide batteriesSecondary batteriesLead/acid batteriesLithium batteriesBattery characteristicsThe futureQuestionsGoing furtherTLP creditsTLP contentsShow all contentViewing and downloading resourcesAbout the TLPsTerms of useFeedbackCredits Print this page
PreviousNext
Voltage delay
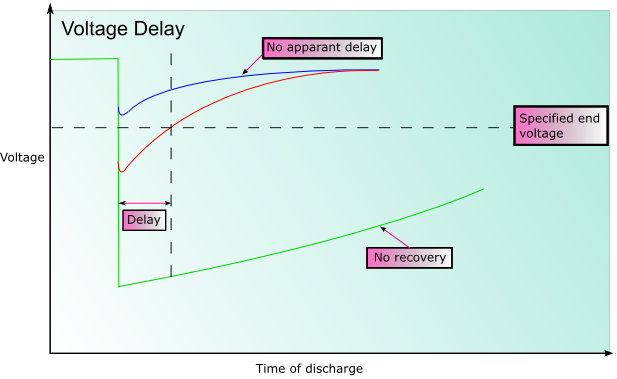
In some battery systems passivation may occur. Passivation is the process by which the reduced product that forms (often an oxide) does not dissolve into the electrolyte, or fall away from the electrode, but forms a film on the surface of the electrode instead. This can significantly impede the reaction as there is reduced electrical contact within the cell. This can substantially prolong the shelf life of the battery, however when the battery discharges the initial voltage may be lower than expected until the coating is broken. This is known as voltage delay.