Experimental matters
Production and measurement of X-Rays
The laboratory source of X-rays consists of an evacuated tube in which electrons are emitted from a heated tungsten filament, and accelerated by an electric potential (typically several tens of kilovolts) to impinge on a water-cooled metal target. When the target’s inner electrons are ejected and outer ones fall to take their place, X-rays are emitted. Some have a continuous distribution of wavelengths between about 0.5 Å and 5 Å ("white radiation") and some have wavelengths characteristic of the electronic levels in the target. For most experiments, a single characteristic radiation is selected using a filter or monochromator.
Methods for obtaining Characteristic Radiation
The diagram below illustrates the characteristic X-ray emission spectrum that is obtained from a copper target.
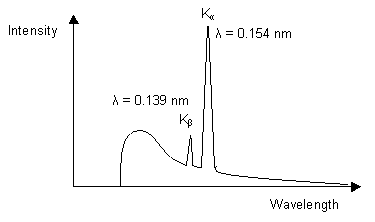
The white and Kβ radiation can be reduced by
1. Using a filter which works by the absorption principle:
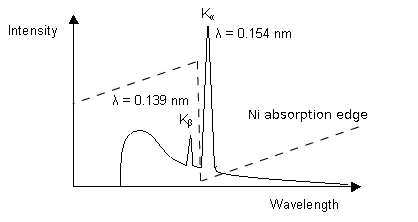
The Ni absorption edge is midway between the Kß and Ka lines so that the former is reduced very substantially while the latter is only marginally reduced.. In selecting the thickness of the filter, a compromise has to be reached between eliminating as much as possible of the undesired radiation and maximising the desired radiation.
For other wavelengths other elements will provide suitable filters.
2. A monochromator that works on the principle of diffraction:
During diffraction a monochromator (a single crystal of known lattice spacing and orientation) is placed in the path of the primary or diffracted beam. The monochromator is set so that the beam is diffracted and only X-rays with the required wavelength reach the detector. (See Bragg’s law).
3. Modern detectors also filter wavelengths (or energy) electronically
Detectors
In the past most X-ray work was done with film, now electronic detectors are used. A single point (e.g. Geiger Muller, scintillation or proportional counter), a line (1D) or an area (2D) detector may be used.

