Tip Surface Interaction
When the tip is brought close to the sample, a number of forces may operate. Typically the forces contributing most to the movement of an AFM cantilever are the coulombic and van der Waals interactions.
- Coulombic interaction: This strong, short range repulsive force arises from electrostatic repulsion by the electron clouds of the tip and sample. This repulsion increases as the separation decreases.
- Van der Waals interactions: These are longer range attractive forces, which may be felt at separations of up to 10 nm or more. They arise due to temporary fluctuating dipoles.
The combination of these interactions results in a force-distance curve similar to that below:
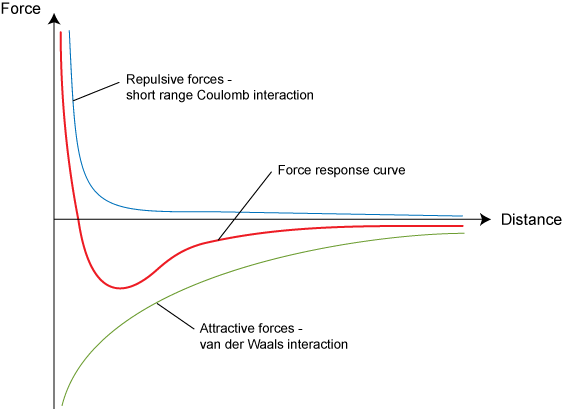
Plot of force against distance
As the tip is brought towards the sample, van der Waals forces cause attraction. As the tip gets closer to the sample this attraction increases. However at small separations the repulsive coulombic forces become dominant. The repulsive force causes the cantilever to bend as the tip is brought closer to the surface.
There are other interactions besides coulombic and van der Waals forces which can have an effect. When AFM is performed in ambient air, the sample and tip may be coated with a thin layer of fluid (mainly water). When the tip comes close to the surface, capillary forces can arise between the tip and surface. These effects are summarised in the animation below.
It is also possible to detect other forces using the AFM, such as magnetic forces to map the magnetic domains of a sample.

