Order and disorder – molecular orientation
Due to their distinctive shape calamitic liquid crystal molecules undergo stronger attractive forces when arranged parallel to one another. They therefore tend to align themselves pointing along one particular direction; this is a known as the director vector and is given the notation n. The angle between individual liquid crystal molecules and the director gives an indication of the orientational order of the system, which can be calculated using the following formula:
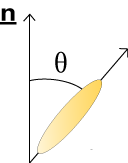 \({\rm{order}}\;{\rm{parameter}}\;Q = {{(3\left\langle{{\cos }^2}\theta \right\rangle - 1)}} \;/\;{2}\)
\({\rm{order}}\;{\rm{parameter}}\;Q = {{(3\left\langle{{\cos }^2}\theta \right\rangle - 1)}} \;/\;{2}\)
When Q = 1 the liquid crystal has complete orientational order; when Q = 0 it has no orientational order and has therefore become an isotropic liquid.
For a thermotropic liquid crystal the variation of Q with temperature follows a trend similar to the one shown in the diagram below (exact values will vary):


