Introduction
Polymers are large molecules made up of long sequences of smaller units. They occur widely in nature and synthetic polymers are a very popular manufacturing material. Polymers are relatively inexpensive to produce on a large scale, and their microstructure can be easily controlled by using different starting materials and by processing them in different ways. This leads to a very wide range of possible properties that are useful for many applications.
The basic structure of a polymer molecule is a long chain of covalently bonded atoms called the backbone. In most common synthetic polymers, the backbone is made of carbon. Attached to the backbone in a regular pattern are other atoms or groups of atoms called side groups. The simplest possible side groups are single hydrogen atoms, as in poly(ethene). The molecules that are bonded together during polymerisation to make up the polymer are called monomers. The arrangement of atoms in the monomer becomes the repeating pattern in the polymer - the repeat unit.
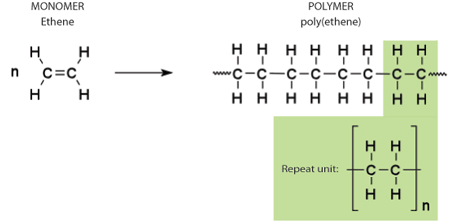
Synthetic examples
You can use the following Flash application to find out about some example synthetic polymers, and to view their monomers. Click and drag the 3D models to rotate them.
Examples of biopolymers
Polysaccharides
Polysaccharides are a type of carbohydrate produced by the polymerisation of individual sugar molecules (monosaccharides). Used as an energy store: starch for plants and glycogen for animals, or for structural support, e.g. cellulose in plants.
Polypeptides
Polypeptides are formed by the polymerisation of amino acids. One or more polypeptide molecules make up a protein. Proteins have a wide variety of functions, which include providing structure and as enzymes, which catalyse chemical reactions.
Collagen is the most abundant protein in the human body, being found in arterial walls, skin, muscle, cartilage, bone and walls of arteries. Amylase is an enzyme which catalyses the digestion of starch, which is a polysaccharide, into its constituent monosaccharides.

