Copper based alloys
Brasses:
Brasses are copper alloys with zinc, see the Cu-Zn phase diagram.
Alpha brass:
From the copper-zinc phase diagram we can see the solid solubility of zinc in copper, for concentrations of zinc upto about 30 at.%, at equilibrium the alloy should be of a single phase.
Alpha brasses are often seen with a single phase, however this usually arises due to annealing. As the alloy cools α phase copper precipitates out first, changing the composition of the remaining melt. This may result in coring and dendritic growth as well as the formation of other phases such as the β phase when the zinc concentration in the remaining liquid is sufficiently high. Annealing the sample to aid diffusion means the composition becomes more uniform as zinc diffuses down the concentration gradient and a single phase predominates.
Alpha-beta brass:
Another very common form of brass is α-β brass. α-β brasses have zinc concentrations of between about 30at.% and 45at.% and are two phase alloys.
The α phase precipitates out first and may form a Widmanstatten structure (see micrograph below), solidifying in plates along preferred growth directions.
Copper-Tin Alloys:
Cu-Sn alloys are sometimes called bronzes, although this includes other kinds of copper alloys (e.g. with silicon and aluminium).
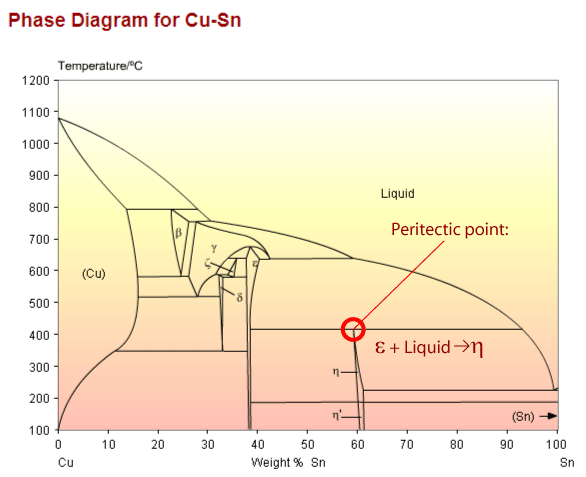
Copper tin phase diagram showing a peritectic point
The peritectic reaction (see diagram above) is an important example of a microstructural transformation. Sn – 21wt.%Cu exhibits this transformation from a solid phase and a liquid phase to a different, solid phase.
Before the transformation begins the system is comprised of the ε phase and liquid. Below 415°C the equilibrium solid phase is η The peritectic transformation begins to take place at 415°C; the new phase precipitates heterogeneously on the surface of ε precipitates. The growing layer of η on the surface of the epsilon precipitates prevents the copper diffusing out to remove inhomogeneities, so some of the copper is trapped within the ε precipitates and the liquid has a lower Cu concentration than the bulk composition. This means the peritectic reaction never goes to completion (i.e. all liquid and solid going to the second solid). In this example the liquid continues to cool until it reaches the eutectic temperature, 227°C, when it transforms.

