Solid oxide fuel cells (SOFCs)
High temperature cells
In the late nineteenth century, conduction was not yet understood. Later, Nernst observed at the University of Göttingen, that stabilized zirconia (ZrO2 doped with Ca, Mg, Y) was an insulator at room temperature, an ionic conductor from 600–1000 °C and a mixed conductor (both electronic and ionic) at around 1500 °C. The main part of the solid oxide fuel cell was therefore discovered. The fuel cell concept was demonstrated by Baur and Preis in the 1930’s using zirconium oxide, but many improvements were necessary to make a competitive device. In the 1950’s, simple, straightforward design made cheaper manufacturing processes possible: the flat plate fuel cell.
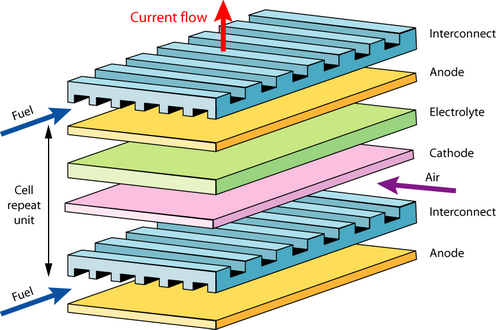
Flat plate solid oxide fuel cell
There are a few problems with the flat plate design when used for larger devices: sealing, around the edges, thermal expansion mismatch and cracking (intrinsically brittle ceramics are used). Tubular designs have been developed to solve these problems (see animation below).
SOFCs are the most efficient devices yet invented, that can convert chemical energy into electrical energy. Both electrodes (cathode and anode) and the electrolyte are made of ceramic materials, since the high operating temperature prevent the use of cheaper metals. The big advantage of the SOFC over the MCFC is that the electrolyte is solid and there are no pumps required to circulate the hot electrolyte. The anode contains nickel, for better electron conduction and catalysis. The operating temperature is between 600 and 1000 °C, depending on the generation of the fuel cell (first, second and third, with decreasing operating temperature). However, thermal cycling can cause cracking of the brittle ceramic components. Both hydrogen and carbon monoxide serve as fuels. Common hydrocarbon fuels can be used in SOFC (diesel, natural gas, gasoline, alcohol etc).
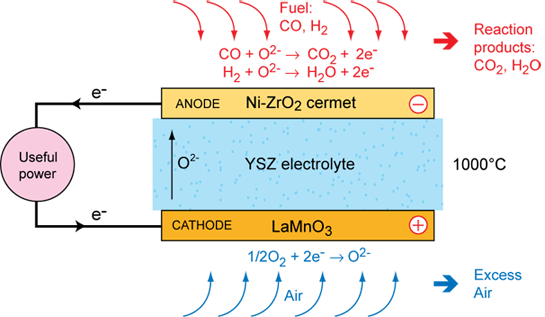
Operation of a SOFC
The operation of the solid oxide fuel cell is straightforward: oxygen atoms are reduced on the porous cathode surface by electrons. The oxide ions diffuse through the electrolyte to the fuel rich and porous anode, where they react with the fuel (hydrogen) and give off electrons to an external circuit. A large amount of heat is produced by the electrochemical reaction, which can be used by an integrated heat management system. Since it takes a long time to reach its operating temperature, the best applications for SOFCs are ones that use both the heat and electricity generated: stationary power plants, auxiliary power supplies. Start-up time problems could be solved by using supercapacitor batteries for the first few minutes of operation in mobile applications.

